Pediatricians' Group Diverges from US Government on Key Vaccine Recommendations
The American Academy of Pediatrics has issued new vaccine recommendations, notably for COVID-19 in children, diverging from U.S. government guidelines for the first time in 30 years amidst differing expert opinions.
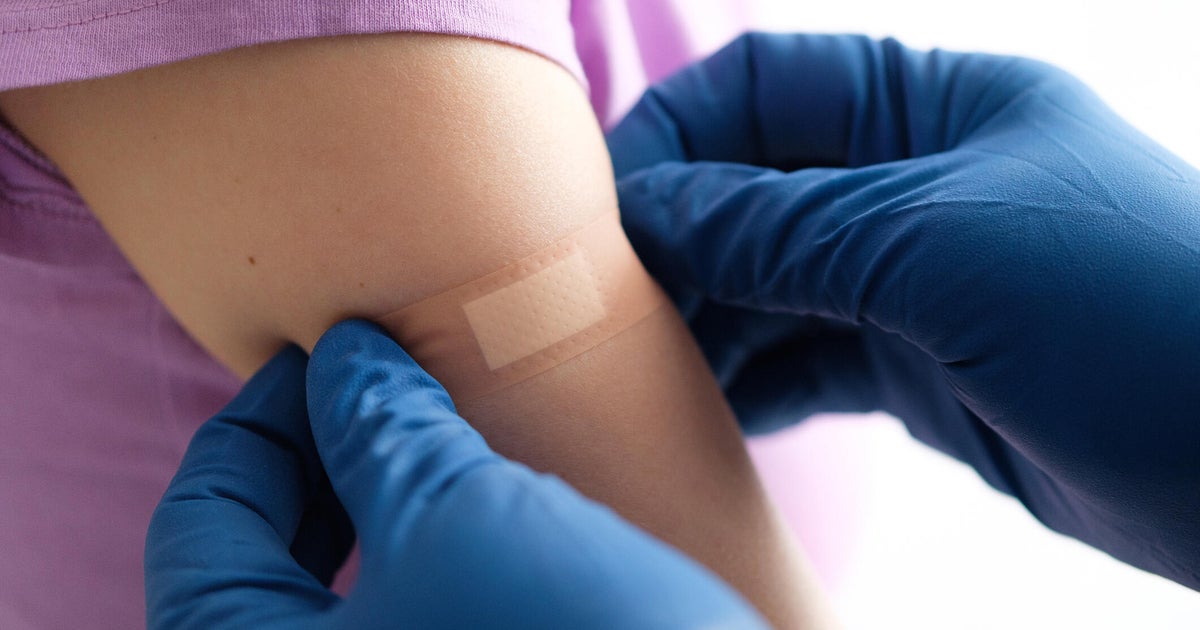
Pediatrics group's COVID-19 vaccine recommendations differ from CDC advice

Leading children's physician group contradicts CDC and recommends COVID-19 vaccines

US pediatricians’ new COVID-19 shot recommendations differ from CDC advice

In break with current CDC recommendations, leading pediatrics group recommends Covid-19 shots for young children
Overview
The American Academy of Pediatrics (AAP) has released new vaccine recommendations, marking its first significant divergence from U.S. government guidelines in 30 years, impacting various immunizations.
The AAP recommends COVID-19 vaccinations for children aged 6 months to 2 years due to their high risk of severe illness, contrasting with U.S. Health Secretary Robert F. Kennedy Jr.'s advice.
Health Secretary Robert F. Kennedy Jr. has advised against COVID-19 shots for healthy children and pregnant women, forming a new panel with vaccine skeptics and excluding major medical organizations.
Differences also extend to flu vaccine advice, with Kennedy's panel recommending thimerosal-free single-dose packaging, while the AAP supports any licensed flu vaccine, citing no evidence of harm.
The AAP also updated guidance for RSV and HPV vaccines, amidst accusations of prioritizing commercial interests due to donations, highlighting a significant shift in public health recommendations.
Analysis
Center-leaning sources frame this story by highlighting the American Academy of Pediatrics' unprecedented divergence from new government vaccine guidance, portraying it as confusing and influenced by 'vaccine skeptics.' They emphasize the AAP's commitment to child protection, while detailing the Health Secretary's controversial actions, suggesting a departure from established public health norms.