CDC Revises Childhood Vaccine Schedule, Narrowing Universal Recommendations to High‑Risk Children
The CDC narrowed childhood vaccine guidance after a White House review, moving several vaccines from universal to risk-based or shared clinical decision-making recommendations for children.
The U.S. models vaccine guidance after Denmark — but forgets the social safety net
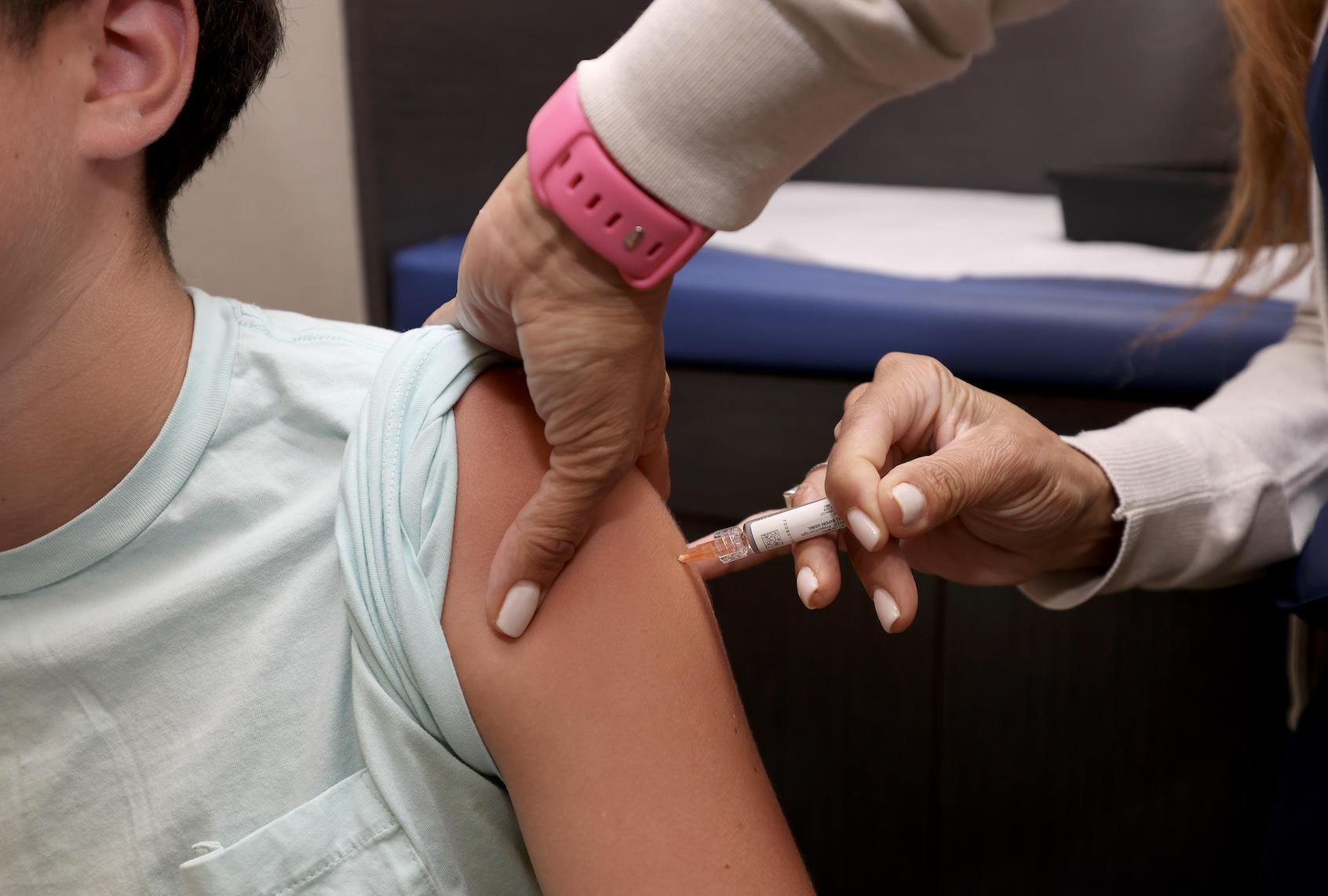
Doctors say changes to US vaccine recommendations are confusing parents and could harm kids

Doctors say changes to US vaccine recommendations are confusing parents and could harm kids
Doctors say changes to US vaccine recommendations are confusing parents and could harm kids
Overview
The CDC this week revised the childhood immunization schedule, removing universal recommendations for hepatitis A and B, rotavirus, RSV, flu, meningococcal and COVID-19 vaccines.
Vaccinations for hepatitis A, hepatitis B, rotavirus, RSV, flu, and meningococcal disease are now recommended only for high-risk children, not universally, per updated CDC guidance.
The change followed President Trump's directive to review U.S. vaccination practices and a White House-ordered review comparing U.S. norms with international standards.
Over 200 health groups urged Congress to act; leading medical societies continue to endorse previous recommendations, and state advisory panels will reassess immunization policies.
Healthcare providers report increased parental inquiries and widespread public misunderstanding since the announcement, with regional differences in vaccination uptake and alternative scheduling choices.
Analysis
Center-leaning sources frame the story by emphasizing the confusion and potential harm caused by the new vaccine recommendations. They highlight concerns from medical professionals about increased vaccine hesitancy and the erosion of trust in science. The narrative is structured around expert opinions and data, portraying the changes as a step back in public health efforts. This framing is evident in the choice of quotes from doctors and the focus on the negative implications of the policy shift.